Are you ready to scale up your business in China?
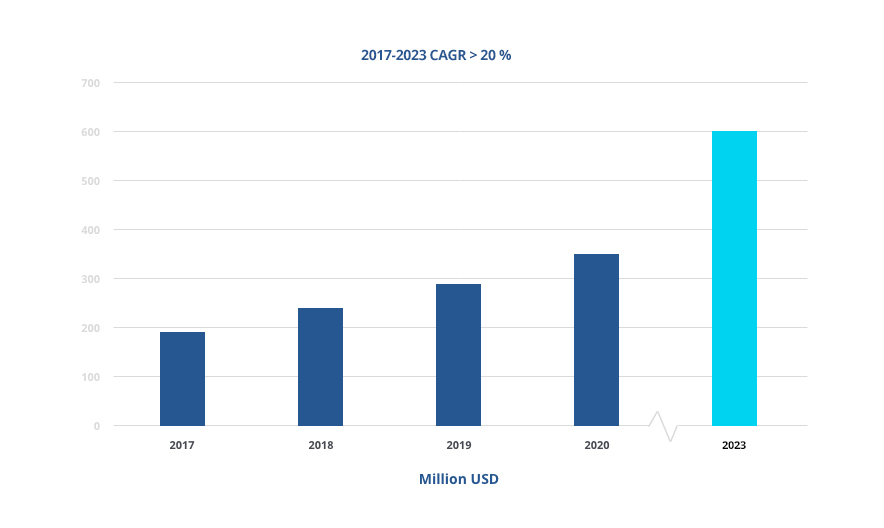
China, a trillion-dollar steadily growing economy is reshaping our global economy. With it, the dental implant market in China is growing rapidly and is estimated to show double-digit growth on average annually between 2020 and 2023, reaching USD 600 million by 2023.
China Dental Implant Market Growth
China-for-China, or manufacturing in China for use in China, has not been used by any global dental implant company - until now.
Historical obstacles include the relatively small market size, a preference for foreign brands, low trust in Chinese quality and the risk of being copied. Moreover, Chinese regulations restrict domestic contract manufacturing of finished products for the Chinese market and clearly limit the traditional use of contract manufacturing.
However, both the size and the mindset of the Chinese market is undergoing tremendous transformations. It’s about time we revise our mindset as well!
New Stronger Incentives for Domestic Production
Savings on import duties, trade tariffs and transportation costs, as well as on registrations, fees and production cost are all excellent reasons to consider domestic manufacturing.
But perhaps even more important incentives are recent initiatives and changes in Chinese regulations:
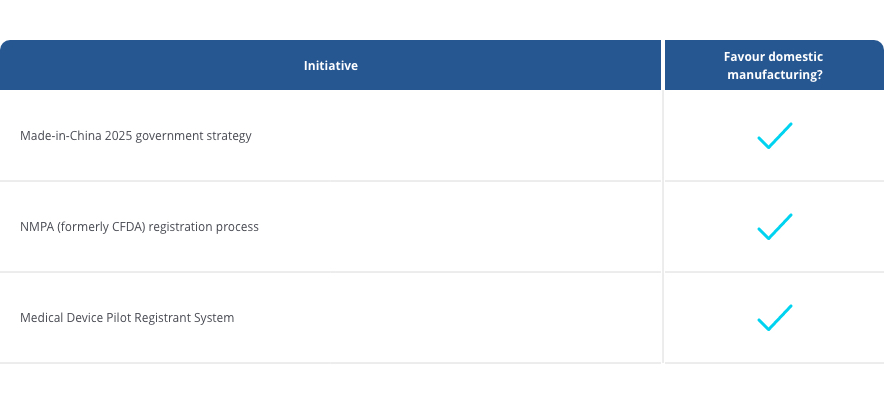
How will these favorable conditions for domestic manufacturers impact sales of foreign manufactured products? Our guess is that it will fuel the demand for domestic manufacturing even further.
The strongest incentive probably continues to be the unstable geopolitical climate endorsed by the present impact and restrictions relating to COVID-19. What will the future bring and how do we handle the uncertainties that we cannot predict or control? Do we have confidence that the geopolitical climate won’t be intensified and impact costs and imports?
You Can Prepare to Scale Up & Shorten Your Time to Market
The historical mindset of domestic manufacturing in China is a thing of the past, and the market is ready to accept the local manufacturing of foreign implant brands.
The Chinese market size and its expected growth, along with the cost and time savings, create great potential for China-for-China. At the same time, avoiding geopolitical uncertainties and potentially shortening time to market.
In the past, Chinese regulations made it impossible for a contract manufacturer to act as intended. The contract manufacturer was forced to take over the same risks as the legal manufacturer and the product owner, as well as be holder of the full technical file.
However, now the Medical Device Pilot Registrant System allows registration applicants, a local enterprise, to apply for medical device approval independently, while only outsourcing production to a qualified contract manufacturer. Neither technical documentation nor registration need to be held by the trusted CMO.
Our Proposal - Contract Manufacturing China-for-China
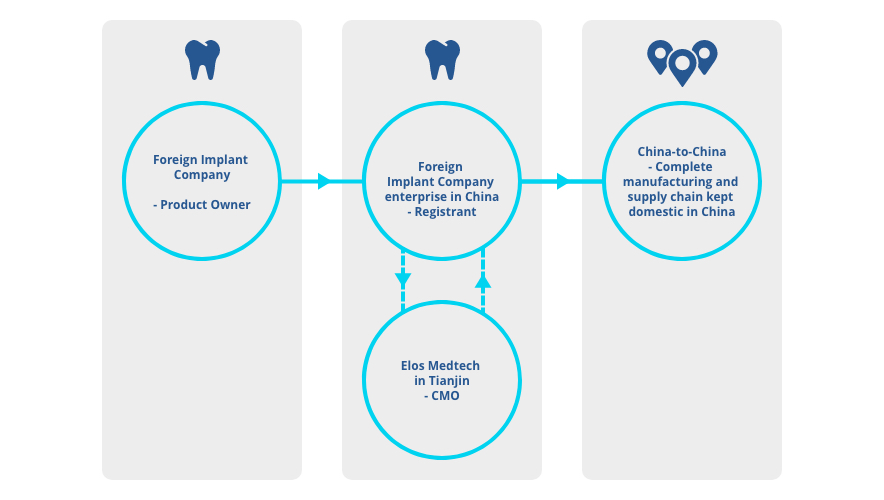
Elos Medtech in China can help you scale up and shorten your time to market contact us for more information.
Elos Medtech in Tianjin, China
Our solution is our Elos Medtech site in Tianjin, your Trusted Partner
- Production according to European standards
- Part of Elos Medtech's global quality system
- ISO 13485 and ISO 14001 certified
- Close collaboration with Elos Medtech Scandinavian dental competence center
- 10 years of medtech experience
- 100% medtech

